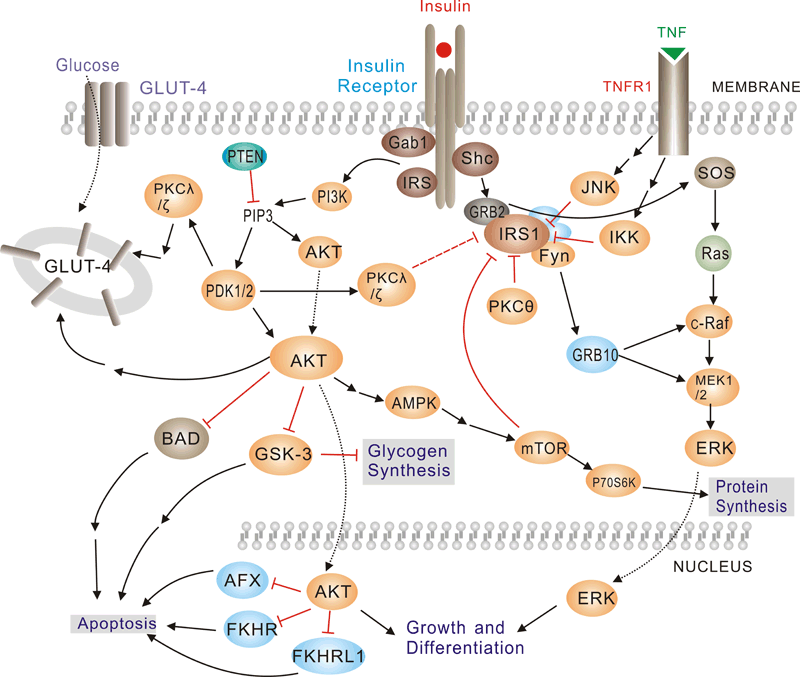
Insulin has diverse effects on cells including stimulation of glucose transport, gene expression and alterations of cell morphology. Insulin signaling begins with either the activation or substrate kinase activity of the insulin receptor (IR), which is the only component of the pathway that is unique to insulin action. The insulin receptor belongs to a subfamily of receptor tyrosine kinases. Activation of the IR can be impaired by post-translational modifications of the protein involving serine phosphorylation, or by binding to inhibiting proteins such as PC-1 or members of the SOCS or Grb protein families. Insulin elicits a diverse array of biological responses by binding to its specific receptor. The hormone mediates these effects by activation of signaling pathways which adaptor molecules such the IRS, the SHC and the GRB2 lipid kinases such as PI3K serine, threonine and tyrosine.
Selected Reviews:
ALAN R,SALTIEL,et al.(2001)Insulin signaling and the regulation of glucose and lipid metabolism.Nature.414(6865):799-806.
Ogawa W,Matozaki T,et al.(1998)Role of binding proteins to IRS-1 in insulin signaling. Mol Cell Biochem.182(1-2):13-22.
Youngren JF. (2007)Regulation of insulin receptor function. Cell Mol Life Sci. 64(7-8):873-91
Johnston AM, Pirola L, Van Obberghen E.2003Molecular mechanisms of insulin receptor substrate protein-mediated modulation of insulin signalling. FEBS Lett. 546(1):32-6.
