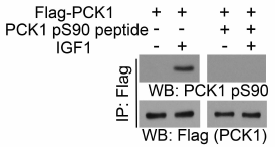
Phosphoenolpyruvate carboxykinase (PCK) is the rate-limiting enzyme of gluconeogenesis in the liver and kidney and converts oxaloacetate and GTP into phosphoenolpyruvate (PEP) and CO2 by adding a phosphate to pyruvate with concomitant aldol cleavage of CO2 from oxaloacetate 1,2. In humans, cytosolic PCK1 shares 63.4% sequence identity with PCK2, which is located in the mitochondria 3. Aberrant PCK expression occurs in many cancers. For instance, PCK1 is overexpressed in melanoma and colorectal cancer, and PCK2 is highly expressed in breast, colon and lung cancer cells4-8. In a recent study, it is reported that AKT in tumor cells phosphorylates cytosolic phosphoenolpyruvate carboxykinase 1 (PCK1) at S909. Phosphorylated PCK1 translocates to the ER, where PCK1 uses GTP as a phosphate donor to phosphorylate Insig1 S207 and Insig2 S151. This phosphorylation reduces the binding of sterol to Insig1/2 and disrupts Insig-SCAP interaction, leading to SCAP/SREBP1 translocation to the Golgi apparatus and subsequent SREBP1 activation and downstream gene transcription for lipogenesis, tumor cell proliferation, and tumorigenesis in mice9.